HomeArticles by: Techsol Life Sciences
Techsol Life Sciences
-
Emerging Trends in Regulatory Affairs by Adapting to Latest Technological Advancements
July 5, 2021 -
Automation Opportunities in Pharmacovigilance
June 25, 2021 -
Global Regulatory Changes in Biopharma
June 21, 2021 -
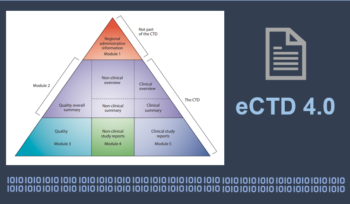
ICH’s eCTD Version 4.0 – Objectives, Major Updates, Resulting Advantages and Possible Challenges
June 14, 2021 -
FDA’s proposed guidance on CSA
May 25, 2021 -
Oracle Argus Safety Application Upgrade
May 10, 2021 -
Combination Products Reporting – Pharma & Medical Devices liaise with NCA
March 3, 2021 -
Factors Influencing the Outsourcing of Pharmacovigilance Processes
January 13, 2021 -
Digital Transformation Catalyst For The Pharmaceutical Industry
October 31, 2020
How can we help you?
Contact us by submitting a business inquiry online. We will get back to you very soon.