-
Literature Screening – Process overview, challenges en-route and the scope for automation
August 30, 2021 -
How Decentralized Clinical Trials Are Expected to Transform the Conduct of Global Clinical Studies
August 5, 2021 -
How the Trial Master File is Expected to Evolve in the Next Three Years
July 28, 2021 -
Emerging Trends in Regulatory Affairs by Adapting to Latest Technological Advancements
July 5, 2021 -
Automation Opportunities in Pharmacovigilance
June 25, 2021 -
Global Regulatory Changes in Biopharma
June 21, 2021 -
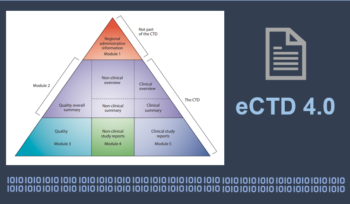
ICH’s eCTD Version 4.0 – Objectives, Major Updates, Resulting Advantages and Possible Challenges
June 14, 2021 -
FDA’s proposed guidance on CSA
May 25, 2021 -
Oracle Argus Safety Application Upgrade
May 10, 2021
How can we help you?
Contact us by submitting a business inquiry online. We will get back to you very soon.